News
Recent News and Stories

- Community
Pinwheels placed at Children’s Hospital of Michigan to raise awareness about child abuse

- Awards and Recognition
Detroit Medical Center Names Josh Hester New CEO for Detroit Campus
- Opthalmology
FOX 2 News: The solar eclipse is exciting, but it can be dangerous - without the right eyewear

- Oncology
Kate Middleton's cancer diagnosis follows months of speculation

- Pediatrics
Doctors alarmed by increased flu-related complications in children across Metro Detroit

- Neurosurgery
12-year-old epilepsy patient celebrated post life-saving brain surgery at CHM

- Pediatrics
Metro Detroit doctors raise concerns over screen time’s impact on children’s health

- Women’s Health
DMC Sinai-Grace Hospital, sorority open ‘Mother’s Closet’ to support new mothers in Metro Detroit
- Infectious Disease
Health department issues warning after Metro Detroit child comes down with highly contagious virus

- News and Articles
DMC expert says pickleball injuries are on the rise

- News and Articles
DMC NICU patients dressed as 'Sweet Valentines' for Valentine’s Day

- Healthy Living
Troy man who collapsed during Belle Isle run thanks those who saved him

- Health and Wellness
How many cups of coffee is too much when it comes to caffeine?
- Diabetes
An accurate diagnosis, treatment for diabetes is a growing concern in pregnancy

- Physical Therapy
It’s a new independence’: Robotic exoskeleton offers new mobility options for paralyzed Detroit woman

- Health and Wellness
DMC Receiving Hospital ER Dr. Trifun Dimitrijevski discusses avoiding frostbite

- NICU
Detroit Welcomes First Baby of the Year at DMC Hutzel Women’s Hospital

- NICU
Babies' first Christmas at DMC Hutzel Women's Hospital & Children's Hospital of Michigan
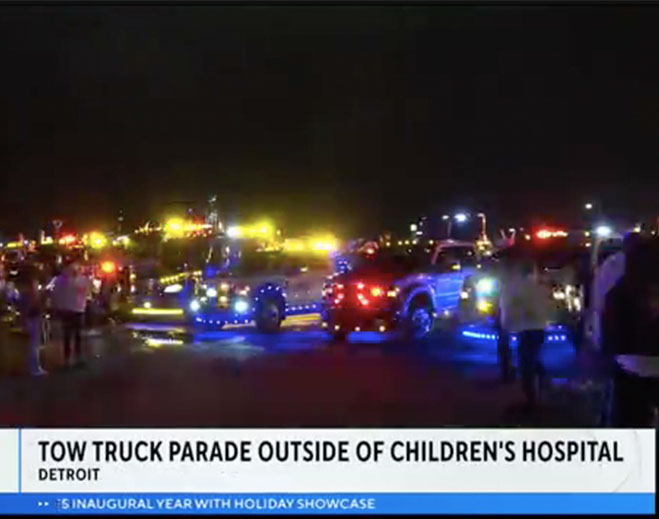
- Community
Tow truck parade takes to streets outside of Children's Hospital of Michigan
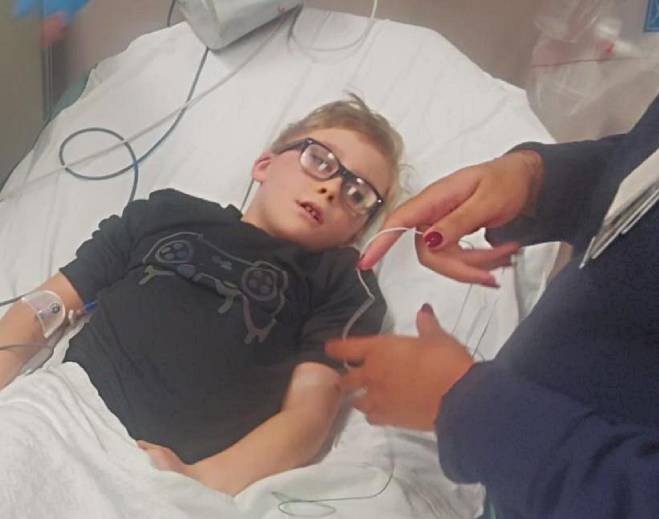
- Pulmonary
DMC expert dispels myth surrounding what is being called White Lung disease

- Pediatrics
How RSV is going to be different this year

- News and Articles
Longtime Michigan community relations professional named
new DMC Group Director of Community Engagement

- News and Articles
Detroit Medical Center names new Chief Nursing Officer
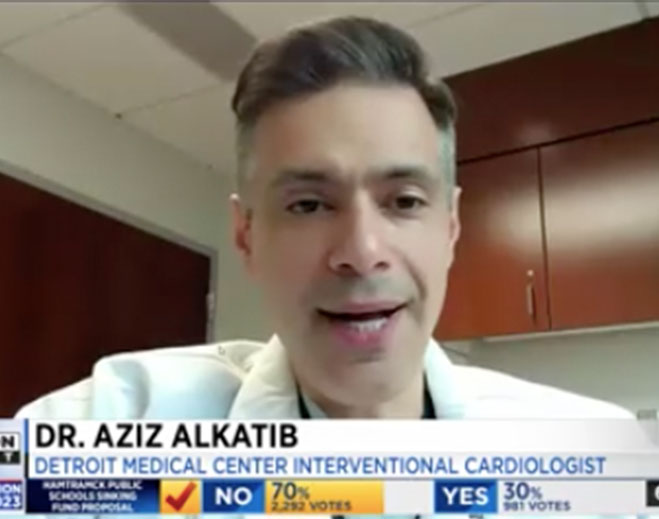
- Cardiovascular
WDIV-TV talks with DMC cardiologist on stress and heart health

- Cardiovascular
WDIV-TV story with DMC cardiologist on research suggesting taking better care of our heart health can help slow down how quickly we age.
- Pulmonary
DMC HVSH expert on robotic-assisted broncoscopy being offered at the hospital

- Health and Wellness
New study looks at the impact and symptoms of long colds and flu

- News and Articles
CHM expert discusses the increase in reported strep throat cases in metro Detroit with WWJ-AM.

- Cardiovascular
DMC cardiologist talked with WWJ-AM about traditional Chinese medicine compound for Heart Attack treatment.

- Awards and Recognition
DMC Leader Recognized on Crain’s 40 Under 40 List

- Neurology
How to identify what's triggering your migraines and ways to manage them as the weather changes

- Sports Medicine
What every pickleball player should know to cut down on injuries
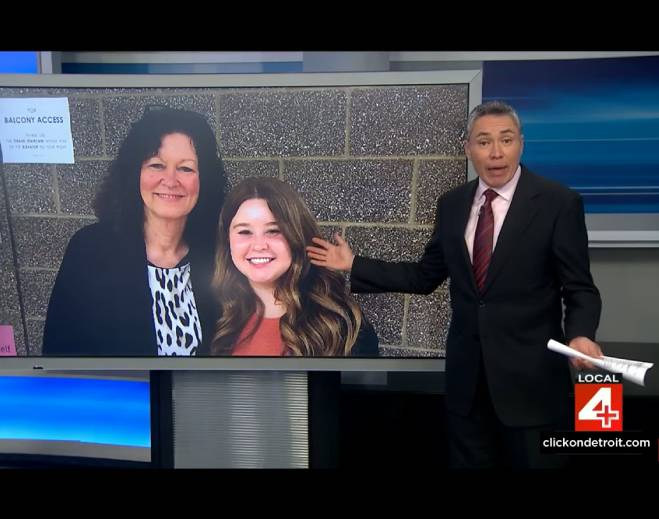
- Oncology
Metro Detroit woman uses her story to bring awareness to breast cancer
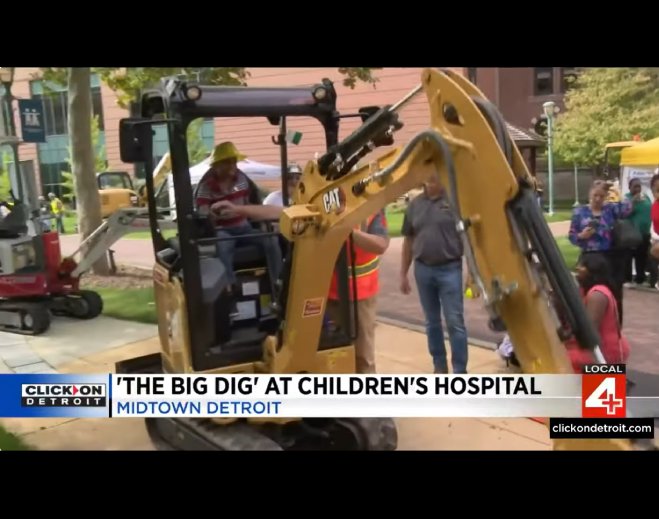
- Community
Children's hospital in Detroit hosts 'Big Dig' event
- Rehab
Nationally recognized rehabilitation center opens site in Southgate

- Orthopedic Rehab
FOX 2 Photojournalist battles back from devastating motorcycle crash
- ENT
DMC Dr. Pulin Patel on ineffective nasal decongestants

- News and Articles
SGH Career Fair

- Awards and Recognition
DMC Rehabilitation Institute of Michigan Recognized on Newsweek’s 2023 List of America’s Best Physical Rehabilitation Centers

- Awards and Recognition
DMC Tenet Heroes 2023

- Nephrology
Clarkston teacher donates kidney to save co-worker's young son
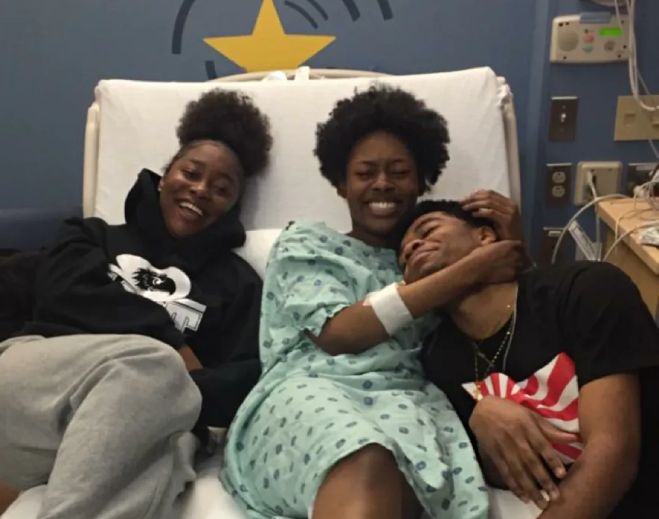
- Nephrology
Kidney recipient pays her ‘Wish’ forward in Metro Detroit

- Cardiovascular
Children's Hospital of Michigan now offering heart surgery clinic in Troy

- Cardiovascular
DMC interventional cardiology expert shares new national guidelines on novel, catheter-based way to access heart

- Orthopedics
2 senior rowers are back on the water after overcoming injuries in Detroit
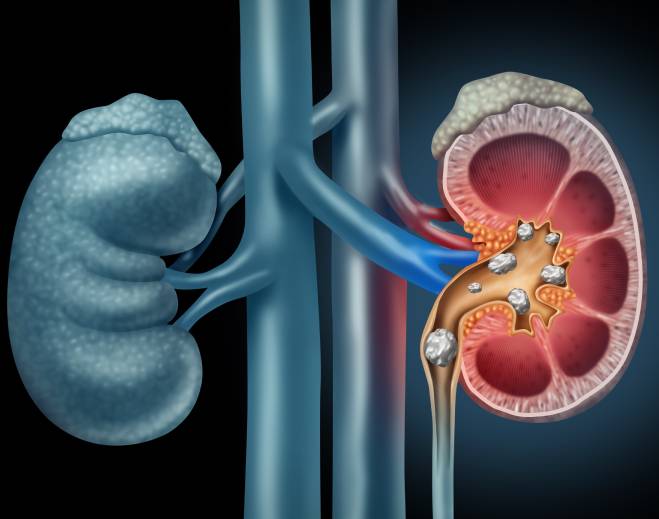
- Nephrology
Kidney stones in kids? It's a growing problem. Here are ways to prevent them

- Health and Wellness
Dr. Kelly Levasseur discusses avoiding heatstroke in a live interview on WWJ-AM

- Community
11-year-old former Children’s Hospital of Michigan patient gives back to kids at facility

- Awards and Recognition
DMC Hospitals Recognized by U.S News & World Report

- Sports Medicine
DMC Sports Medicine expert Dr. Christopher Cooke is featured in this story on WJR Health Watch on concussions

- Cardiovascular
Dr. Nasser Lakkis was interviewed by WWJ-AM on Lebron James’ son’s cardiac arrest

- Health and Wellness
Weekend warrior or daily workouts: study seeks best time for fitness
- Health and Wellness
Health expert discusses how to stay safe during tick season

- News and Articles
Teacher donates lifesaving gift to colleague’s son

- Awards and Recognition
DMC Hospitals Ranked Nationally for Stroke Care

- Pediatrics
How a $70 device helped a mother save her 1-year-old son from choking on toy

- Men’s Health
Why don't men go to the doctor as often as women?

- Health and Wellness
Doctors are sounding the alarm about malaria in the U.S.

- Health and Wellness
Here’s how to protect kids from top summer injuries in Metro Detroit

- News and Articles
Smoke from Canadian wildfires creates respiratory hazards for southeast Michigan residents

- Pediatrics
Advocacy group joins doctors to help families of children with asthma on World Asthma Day
- Pediatrics
What a new study found about the pandemic’s impact on mental health

- Pediatrics
Kindergartner dies amid an outbreak of unidentified illnesses at Detroit school

- News and Articles
How artificial intelligence is saving lives in our hospitals

- Announcement
New CEO at Children’s Hospital of MichiganArchie Drake named new CEO at Children's Hospital of Michigan

- News and Articles
Metro Detroit teen starts foundation to provide rides to sickle cell clinic for children
- News and Articles
Deadly drug-resistant infections caused by ‘Candida Auris’ are rising in Michigan hospitals

- Pediatrics
Meet the '313 Day' newborns at the Detroit Medical Center

- Announcement
New CEO at Children’s Hospital of Michigan

- Cardiovascular
What you need to know about heart attacks as we wrap up Heart Health Month
- Pediatrics
Metro Detroit family creates lifesaving plan for baby with heart defect

- Announcement
New CEO at DMC Huron Valley-Sinai Hospital
- News and Articles
DMC Detroit Receiving Hospital Burn Center Re-verified

- Announcement
New Designated Institutional Officer at the Detroit Medical Center

- Health and Wellness
WWJ: If you think Seasonal Depression is just about cold weather and gray skies, think again

- Health and Wellness
WWJ: Sitting Too Much Is Not Good For Overall Health

- Announcement
DMC smart watch giveaway includes cookbook with entry

- News and Articles
Sunday scaries. Revenge bedtime procrastination. Work Stress. Why don't Michiganders get enough sleep?

- Announcement
Detroit Medical Center Announces New Specialist-in-Chief of Orthopedics

- Health and Wellness
Dr. Chadi Alraries, Cardiologist at the Detroit Medical Center, helps shed light on Damar Hamlin's collapse

- Health and Wellness
New Year’s babies: Metro Detroit hospitals announce first babies born in 2023

- Health and Wellness
Meet the first babies born in the New Year in metro Detroit

- Health and Wellness
WDIV: Keeping Track of Your Blood Pressure During the Holidays
- Health and Wellness
Here's what you need to know to prevent carbon monoxide poisoning in your home

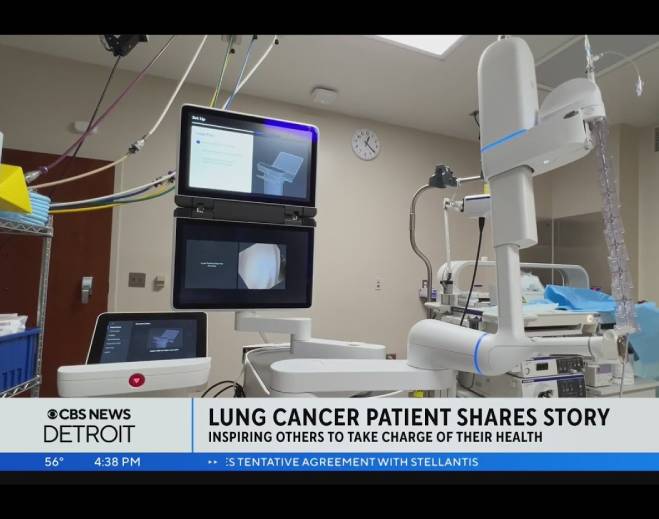
- Lung Cancer
Simple lung cancer screening test is easy, painless — and could save your life

- News and Articles
Valentini: Spread cheer, not illness this holiday season. Get vaccinated | Opinion

- News and Articles
WWJ: How to Avoid Those Extra Pounds During the Holidays

- News and Articles
Getting less than five hours of sleep raises risk of chronic diseases

- News and Articles
Dale Lantzer from Kalkaska Climbed Mount Kilimanjaro after Several Setbacks

- Health and Wellness
WWJ Your Eye on Health: Should You Keep Your Blood Pressure Under Control?

- News and Articles
Metro Detroit father battling extremely rare case of West Nile virus

- Health and Wellness
WWJ Your Eye on Health: Officials Urge the Public to Get Their Flu Shots

- News and Articles
Children’s Hospital of Michigan study shows steroid treatment improves survival of preterm infants

- News and Articles
Detroit food truck owner learns to walk again, gets back to work after being intentionally run over
- Announcement
National Brain Injury Grant Awarded to Wayne State University and DMC Rehabilitation Institute of Michigan

- Awards and Recognition
Crain's 40 Under Forty: Brittany Lavis, 32

- Health and Wellness
WWJ Your Eye on Health: Increased Alcohol Consumption Tied to Increased Risk of Cancer
- Announcement
DMC Rehabilitation Institute of Michigan Welcomes New Chief Nursing Officer

- Women’s Health
DMC Names New Specialist-in-Chief of Obstetrics and Gynecology

- Awards and Recognition
DMC Rehabilitation Institute of Michigan Ranked Among the Best in the U.S. on Newsweek's 2022 List of America's Best Physical Rehab Centers

- Health and Wellness
FOX 2 News: DMC takes part in “Health is Wealth” event

- Announcement
WWJ-AM: DMC’s CHRO was interviewed about an employee recruitment event

- Cardiovascular
WWJ Your Eye on Health: Advice on At-Home Devices for Blood Pressure

- Hospital Administration
DMC Sinai-Grace Hospital Announces New CEO

- Hospital Administration
DMC Announces New CEO for Sinai-Grace Hospital

- News and Articles
Michigan couple marries at Sinai-Grace Hospital after bride's mom is placed on life support
- Announcement
DMC Huron Valley-Sinai Hospital Offering Robotic-Assisted Minimally Invasive Option For Lung Biopsy
- Health and Wellness
Fighting plantar fasciitis: How to ease common foot pain

- Neurology
WWJ Your Eye on Health: Symptoms of Depression May Be a Warning Sign for Stroke

- Awards and Recognition
DMC Hospitals Recognized for Care

- Hospital Administration
New CEO Announced at DMC Sinai-Grace Hospital

- Cardiovascular
WWJ Your Eye on Health: Cancer Survivors at Higher Risk for Heart Disease

- Hospital Administration
Brittany Lavis Joins Michigan Health & Hospital Association Board

- Cardiovascular
WWJ Your Eye on Health: Vitamin Supplements Not Found to Prevent Heart Disease or Cancer

- Cardiovascular
WWJ Your Eye on Health: Too Much Sitting Can Put You at Risk for Heart Disease

- Awards and Recognition
DMC Hospitals Nationally Recognized for High Quality Stroke Care

- Tenet Heroes
Detroit Medical Center Names Tenet Heroes

- Stroke
DMC Sinai-Grace Hospital Nationally Recognized for High Quality Stroke Care

- Health and Wellness
WWJ Your Eye on Health: Smiling Will Make You Feel Good

- Gastroenterology
WWJ Your Eye on Health: Laparoscopic Ulcer Surgery Leads to Dramatically Shorter Hospital Stays
- Community
Laparoscopic Ulcer Surgery Leads to Dramatically Shorter Hospital Stays and Quicker Recovery

- Obstetrics
Romper: Breaking Down The Different Types Of Postpartum Infections

- Community
WXYZ: Moms showing up empty-handed at hospitals get help from local sorority

- Community
WWJ Your Eye on Health: Use Humor as Primary Flirting Technique

- Community
WWJ Your Eye on Health: Parenting Burnout Taking its Toll on Two-Thirds of Parents
- Oncology
WXYZ: Women's Health Week Puts Spotlight on Breast Health

- Community
DETROIT MEDICAL CENTER CELEBRATES NATIONAL HOSPITAL WEEK

- Community
DMC Honors National Nurses Week

- Orthopedics
WWJ Your Eye on Health: Shorter Recovery Time Possible for ACL Injuries

- Cardiovascular
WWJ Your Eye on Health: New Guidelines for Aspirin Use to Decrease Risk of Heart Disease

- Health and Wellness
WWJ Your Eye on Health: Flu Cases on the Rise in Detroit Area

- Cardiovascular
WWJ Your Eye on Health: Liver Condition in Adults May Lead to Heart Disease

- Allergies
WWJ Your Eye on Health: History of Allergies May Be At Risk of High Blood Pressure
- Orthopedics
DMC Huron Valley-Sinai Hospital first in Michigan to Offer Ultrasound Guidance Device for Trigger Finger Treatment

- Community
WWJ Your Eye on Health: Avoiding the Spring Break Hangover After Vacation

- Sports Medicine
WWJ Your Eye on Health: Sports Injures Are On The Rise; Reduce Your Risk

- Emergency
WWJ Your Eye on Health: When to Seek Emergency Care for Spring Allergies

- Hospital Administration
Detroit Medical Center Welcomes New Specialist-In-Chief of Neurology

- Sleep
Should we stay on daylight saving time? Debate goes beyond eliminating seasonal time changes

- Cardiovascular
WWJ Your Eye on Health: Springing Forward Can Lead to Health Problems

- NICU
Meet DMC hospital's 'Born in the 313' babies on 313 day!

- Health and Wellness
WWJ Your Eye on Health: Spending Too Much Time on Your Devices Can Be Unhealthy

- Oncology
WWJ Your Eye on Health: What to Know About Colon Cancer Awareness Month

- COVID-19
WWJ Your Eye on Health: Recent Report Shows Decrease in Anxiety for Those Who Are Vaccinated Against COVID-19
- Community
DMC Videos Celebrate Black History Month

- Awards
DMC Sinai-Grace Hospital Earns ACR Accreditation

- Awards and Recognition
HVSH Recognized as one of West Oakland County’s Best

- Hospital Administration
DMC Sinai-Grace Hospital Names New Chief Nursing Officer

- Cardiac
WWJ Your Eye on Health: Simple Tips for Shoveling Snow

- COVID-19
WWJ Your Eye on Health: Area Hospitals Seeing Drop in COVID-19 Patients

- Emergency
WWJ Your Eye on Health: Close Look at What Emergency Departments Have Seen During Snowstorm

- Community
MyTV20: Dangers of Snow Shoveling

- Hospital Administration
Chief Executive Officer Named at the Detroit Medical Center
- Community
LGBTQ+ community, supporters call FDA policy on donating blood 'discriminatory' amid shortage

- COVID-19
Young males at increased risk for inflammation of heart muscle after COVID booster: study
- COVID-19
Examining efforts at hospitals to stay ahead of the COVID-19 surge

- COVID-19
Amid Covid Spike, Health Experts Recommend Small New Year’s Eve Celebrations

- COVID-19
Patients wait hours for emergency care as Michigan hospitals overwhelmed by COVID-19

- COVID-19
Health Officials Advise Surgical Masks Over Cloth To Protect Against Spread Of COVID-19

- Community
WJR: How Do You Manage All of the Stress During the Holidays?

- COVID-19
With Omicron detected in Michigan, doctors and people are concerned as holidays are around the corner
- Health and Wellness
Pandemic stress, work stress, now holiday stress; Here are some ways to cope

- COVID-19
WWJ Your Eye on Health: Could All Adults Soon Be Eligible for a COVID-19 Booster Shot?

- COVID-19
Taylor man declined COVID-19 shot and nearly died. 'Get vaccinated ... it's a real disease'
- COVID-19
Detroit's Renaissance High School turns to remote learning after COVID-19 outbreak
- COVID-19
Michigan man survives grueling battle against COVID-19 thanks to DMC team and ECMO

- Hospital Administration
DMC Names New Group Chief Operating Officer
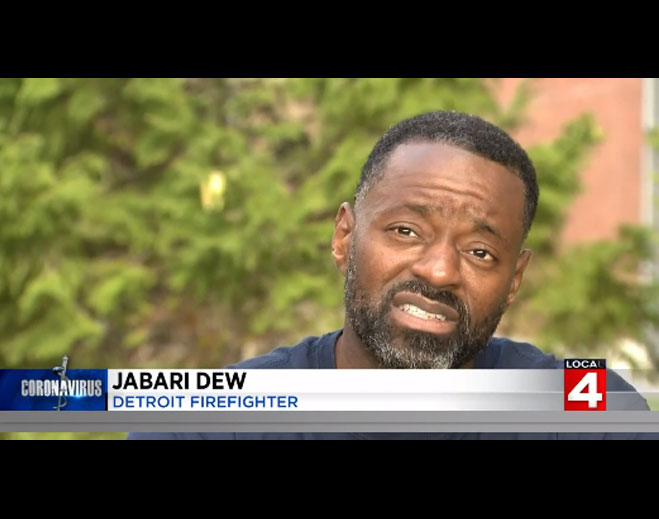
- COVID-19
Detroit firefighter works to get healthy after contracting COVID

- Community
Crain's 40 Under Forty: Jacqueline (Gennie) Snow

- Health and Wellness
WWJ Your Eye on Health: Young Adults Twice as Likely to Turn to Cannabis to Manage Pain

- Hospital Administration
DMC Huron Valley-Sinai Hospital Earns Level III Trauma Center

- Stroke
WWJ Your Eye on Health: Low Dose Aspirin Not Advised To Prevent Heart Attack or Stroke

- Health and Wellness
WJR: Baby Aspirin Not Recommended for Older Adults

- Health and Wellness
WWJ Your Eye on Health: Internet Gaming Disorder Affects 1 in 4 College Students

- Oncology
WWJ Your Eye on Health: Get Screened For Colon Cancer

- Mammogram
WWJ Your Eye on Health: Reminder of Importance of Screening Mammograms

- Cardiovascular
WWJ Your Eye on Health: Half of Adults in U.S. Have Uncontrolled High Blood Pressure

- COVID-19
WWJ Your Eye on Health: CDC Urges Pregnant Women to Get Vaccinated Against COVID-19

- Community
September Brings Mental Health to the Forefront

- Awards
Dr. Arti Madhavan Named Specialist-in-chief of Family Medicine at the Detroit Medical Center

- Community
'We're young and dumb': The milk crate challenge is the latest internet trend you probably shouldn't try

- Hospital Administration
Kimberly Ronnisch Named Chief Nursing Officer at DMC Adult Central Campus

- Spinal Cord Injury
College senior suffers severe spinal injury, unlikely to walk - makes remarkable recovery
- COVID-19
Physician & Nurse Leaders from 21 Hospital Systems Across Michigan Plead for Vaccinations

- Awards and Recognition
DMC Rehabilitation Institute of Michigan Ranked #1 in Michigan on Newsweek's America's Best Physical Rehab Centers 2021 List

- Awards and Recognition
DMC Huron Valley-Sinai Hospital Physician Named Emergency Physician of the Year

- Health and Wellness
WWJ Your Eye on Health: Tips for Staying Safe in Extreme Heat

- Sports Medicine
WJR: How Athletes Can Practice Safely in Extreme Heat

- COVID-19
Detroit Medical Center Updates Visitor Policy

- Community
Sinai-Grace Guild CDC Launches Live Local Program in Northwest Detroit

- Maternity
WXYZ: Summer Heat Safety Tips for Pregnant Women

- Awards and Recognition
DMC Hospitals Ranked Nationally for Care

- COVID-19
WWJ Your Eye on Health: Health Problems Linger for Some COVID-19 Patients

- Community
WWJ Your Eye on Health: How to Avoid Certain Injuries While Running

- Community
Community Partner Spotlight

- COVID-19
WWJ Your Eye on Health: Dr. Audrey Gregory Offers Lessons Learned During the Pandemic

- Health and Wellness
WWJ Your Eye on Health: Health of Your Heart Influenced by Wealth

- Health and Wellness
WWJ Your Eye on Health: Avoid the Dangers of Flood Waters

- Community
DETROIT MEDICAL CENTER UPDATES VISITOR POLICY - July 2nd

- Community
WWJ Your Eye on Health: Detroit Receiving Hospital Doctor Takes Care of Everyone at Grand Prix

- Health and Wellness
WWJ Your Eye on Health: Return to Work Creates Anxieties Among Workers
- Community
WXYZ: What you need to know about fireworks hazards and safety heading into the holiday weekend

- Obstetrics
Sinai-Grace Hospital Delivers a Personalized Birthing Experience

- Awards
DMC Huron Valley-Sinai Hospital's Maternal Health Program Honored

- Health and Wellness
WebMD: Meeting Couples' Different Sleep Needs

- Community
Scholarship Available for Students Attending DMC Sinai-Grace Hospital School of Radiologic Technology

- Community
DMC Official Medical Provider for Grand Prix

- Awards and Recognition
DMC Sinai-Grace Named Most Racially Inclusive Hospital in State

- Awards and Recognition
Crain's Health Care Heroes: Teena Chopra, M.D., Director of Infection Prevention and Hospital Epidemiology

- Oncology
WWJ Your Eye on Health: Get Screened For Colon Cancer Earlier

- Health and Wellness
WWJ Your Eye on Health: How to trim the pandemic pounds

- Awards and Recognition
Dr. Audrey Gregory named one of Crain's Detroit Business' 2021 Notable Executives in Diversity, Equity and Inclusion

- COVID-19
WXYZ: Long-Term Symptoms Impacting COVID-19 Survivors
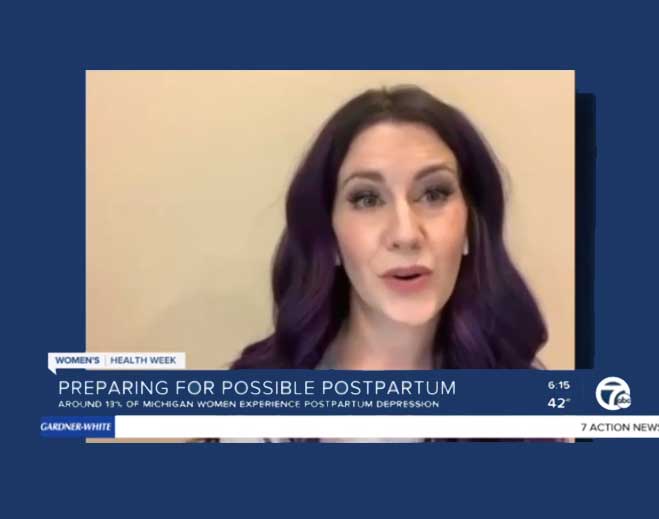
- Women’s Health
Women's Health Week: Everything you need to know about postpartum health

- Community
Daughter follows in mother’s footsteps in career at Detroit Medical Center

- Health and Wellness
WJR Paul W. Smith Show: Lack of Sleep in Middle Age May Lead to Dementia

- COVID-19
WJR Paul W. Smith Show: COVID-19 Vaccines Are Safe For Pregnant Women

- Maternity
WWJ Your Eye on Health: Are the COVID-19 Vaccines Safe for Pregnant Women?

- COVID-19
WWJ Your Eye on Health: Detroit Woman Returns Home After COVID-19 Hospital Stay

- COVID-19
WJR Healthwatch: Recovering COVID Patients Face Long Therapy

- Community
DMC Detroit Receiving Hospital Reverified as Level I Trauma Center

- Tenet Heroes
Six DMC Team Members Named Tenet Heroes

- COVID-19
COVID-19 Visitor Restrictions for Inpatients

- Oncology
WWJ Your Eye on Health: Don't Let the Pandemic Keep You From Cancer Screenings

- Health and Wellness
WYXZ: Taking Control of Your Excess Weight During the Pandemic

- Health and Wellness
Should you supplement your diet with immune-supportive vitamins during the pandemic?
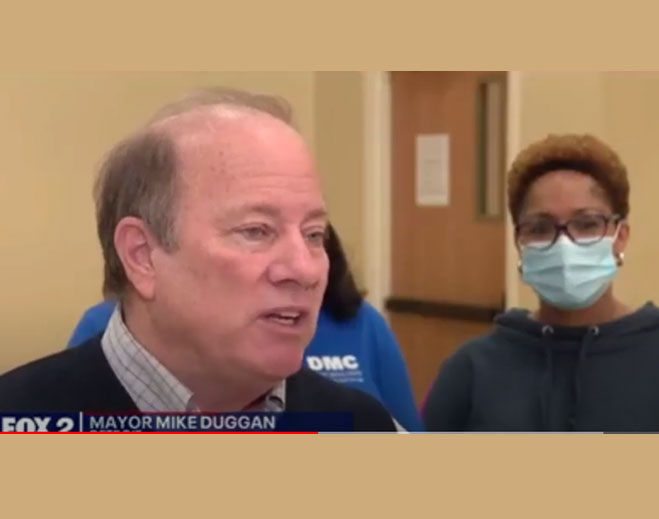
- COVID-19
Mayor Duggan Visits Mass Vaccination Site
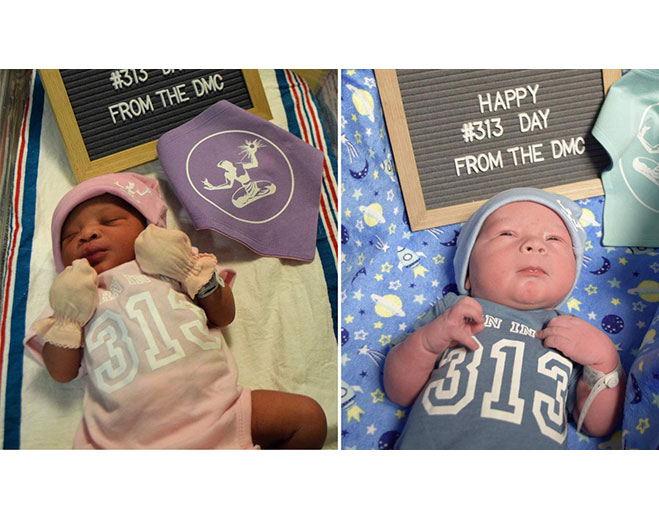
- Maternity
Newborn babies celebrate 313 Day with Detroit apparel at DMC hospital

- Health and Wellness
WJR Paul W. Smith Show: Time Change Affects People In More Ways Than One

- Oncology
WWJ Your Eye on Health: What to Know About New Lung Cancer Screening Guidelines

- Health and Wellness
WWJ Your Eye on Health: Pay More Attention To Your Diet Than Your Workout

- COVID-19
WWJ Your Eye on Health: Detroit Man Battles Back From COVID-19

- Gastroenterology
WWJ Your Eye on Health: Colon Cancer Screening More Important Than Ever
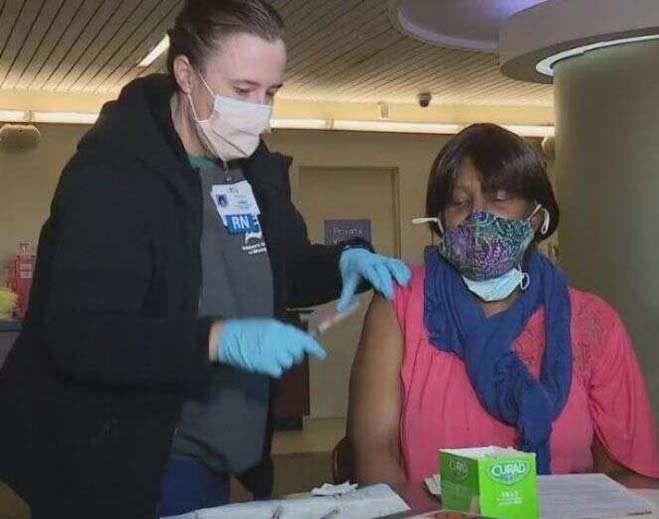
- COVID-19
DMC partners with Detroit area churches for COVID-19 vaccinations

- COVID-19
WWJ Your Eye on Health: Pre-existing Conditions and COVID-19

- Orthopedics
WJR's Paul W. Smith Show: Traumatic Leg Injuries and the Long Road to Recovery

- Women’s Health
WWJ Your Eye on Health: Women and Heart Disease

- Community
Detroiter becomes first Black woman to become chief of neurosurgery at DMC

- Cardiovascular
Detroit surgeon saves life of 85-year-old South Lyon man other hospitals turned away

- COVID-19
Dearborn teachers line up for COVID vaccines at DMC

- Community
Local doctor becomes first Black woman to lead neurosurgery department at Detroit Medical Center

- Cardiovascular
Mason Minute: Dr. Kaushik Mandal on the Importance of Cardiac Care

- Cardiovascular
WWJ Your Eye on Health: How Cold Weather Affects Your Heart Health

- Cardiovascular
WWJ Your Eye on Health: Negative Health Effects of Being Overweight

- COVID-19
WWJ Ask a Doctor: Second Doses of COVID-19 Vaccine Being Distributed

- Health and Wellness
WWJ Ask a Doctor: Types of Mental Health Issues During the Holidays

- Health and Wellness
WWJ Your Eye on Health: Tips for a Healthier You in 2021
- Community
WXYZ: Substance Abuse on the Rise in Metro Detroit
- COVID-19
Detroit mother of 5 returns home after delivering twins while battling COVID-19
- COVID-19
Spending Christmas with family is next goal for recovering COVID-19 patient
- Surgery Center
DMC Huron Valley-Sinai Hospital Offers Micro-Invasive Option for Carpal Tunnel Syndrome

- News and Articles
Detroit Medical Center Names New Group Chief Nursing Officer

- COVID-19
WWJ Ask a Doctor: Mental Health Issues on the Rise During COVID-19 Pandemic

- Community
DMC Names Specialist in Chief of Neurosurgery

- News and Articles
DMC Sinai-Grace Pioneering Unique Robotic Bladder Surgery

- Gynecology
DMC Huron Valley-Sinai Hospital first in State to Use Incision Free Procedure for Uterine Fibroids

- COVID-19
WWJ Ask A Doctor: How to Best Protect Yourself From COVID-19 During Thanksgiving

- Health and Wellness
WJR's Paul W. Smith Show: Don't Delay Your Care Due to COVID-19

- Women’s Health
WWJ: Common Reasons Women Experience Miscarriages
- Health and Wellness
DMC Medical Matters | Hospital safety and not delaying your care

- COVID-19
WWJ Ask A Doctor: What Detroiters should do in the face of a second wave of COVID-19
- COVID-19
Detroit area hospitals confident in handling surge of COVID-19 as hospitalizations soar

- Hospital Administration
Detroit Medical Center Hires New Chief Medical Officer for Adult Central Campus Hospitals

- COVID-19
WWJ Your Eye on Health: Warren man has words of advice while rehabbing from COVID-19

- Community
DMC UPDATES VISITOR RESTRICTIONS

- Awards and Recognition
A Video Profile of Dr. Audrey Gregory, One of Modern Healthcare's Top 25 Minority Leaders

- COVID-19
CEO Dr. Audrey Gregory
- Oncology
WXYZ: Mammograms & self checks for breast cancer are a must even amid pandemic

- COVID-19
WXYZ: Detroit man with Down syndrome faces long recovery after battle with COVID-19

- COVID-19
WWJ Your Eye on Health: Alcohol Consumption On The Rise During Pandemic

- Health and Wellness
WJR Paul W. Smith Show: Importance of Getting a Flu Shot This Year

- Health and Wellness
DETROIT MEDICAL CENTER FOCUSING ON UPCOMING FLU SEASON

- Health and Wellness
WWJ Your Eye On Health: Easing Your Way Back Into The Gym

- Hospital Administration
DMC Names Neurosurgery Leader at Harper University Hospital

- Colorectal
WJR's Paul W. Smith Show: Screening for Colon Cancer More Important Than Ever
- Colorectal
MyTV 20: Importance of Colon Cancer Screening

- Oncology
WWJ Your Eye on Health: Colon Cancer Screening More Important Than Ever

- Community
White Lake resident recognized for nursing excellence, named Exemplary Citizen
- COVID-19
MyTV20: Major Push for Flu Shots in Metro Detroit

- Hospital Administration
DMC Rehabilitation Institute of Michigan Appoints Assistant Chief Nursing Officer

- COVID-19
Best practices for avoiding coronavirus 6 months into the pandemic

- Health and Wellness
WWJ Your Eye on Health: Safely Setting Up A Home Office

- COVID-19
Coronavirus (COVID-19) survivor heads home after 73 days on ventilator in Detroit hospital

- Hospital Administration
DMC Names New CEO for Adult Central Campus Hospitals

- Emergency
WWJ Your Eye on Health: Emergency Departments Are Safe and Open

- COVID-19
WWJ Your Eye on Health: COVID-19 Linked with Obesity

- Awards and Recognition
DMC Hospitals Ranked Nationally for Care

- Community
WJR Paul W. Smith Show - Boating and water safety

- COVID-19
WWJ Your Eye on Health - COVID-19 Rehabilitation Recovery Program

- COVID-19
- Psychiatry
WWJ Your Eye on Health - What is Doomscrolling?

- Community
One minute interview with DMC Huron Valley-Sinai Hospital CEO David McEwen

- Oncology
WWJ Your Eye on Health - HPV Vaccine Guidelines

- Cardiovascular
WWJ Your Eye on Health - Lowering the risk of Heart Disease
- Cardiovascular
WWJ Your Eye on Health: Increase in Broken Heart Syndrome

- Health and Wellness
WJR Paul W. Smith Show: Staying Safe in the Heat
- COVID-19
Identifying and Preventing COVID-19 at Skilled Nursing Facilities

- COVID-19
Large jump in Michigan deaths outside hospitals as coronavirus raged

- Community
How Michigan auto insurance savings could prove costly if you’re seriously hurt in a crash

- Community
Dr. Audrey Gregory Joins Michigan Health & Hospital Association Board

- Community
Michigan's New Auto No-Fault Insurance Law

- COVID-19
DMC Eases Visitor Restrictions

- Community
River Rouge high schooler celebrates surviving COVID-19 after 3 weeks on ventilator

- COVID-19
Rehabilitation Institute of Michigan Launches COVID-19 Rehab Recovery Program

- Emergency
Detroit Medical Center Shows Its Appreciation During National Emergency Medical Services Week

- Community
Virtual Cereal Drive Because Now More than Ever, Kids Need Breakfast

- Community
DMC Children’s Hospital of Michigan Names New CEO
- Community
COVID-19 survivor shares his battle to live after 6 weeks of hospitalization: I am a miracle

- Hospital Administration
Detroit Medical Center Celebrates National Nurses Week 2020

- Community
Dr. Gregory Invites You to Hear from the Sinai-Grace Front Line

- Community
DMC CEO Community Message
- Health and Wellness
DMC adaptive yoga aids those ill or with injuries

- Health and Wellness
COVID-19 Facts for Persons with Disabilities

- Hospitals
Rehabilitation Institute of Michigan Relocates Some Appointments

- Awards and Recognition
Patty Jobbitt Named as One of Crain's Detroit Business Most Notable Women in Healthcare

A Memo to our Patients

What Is an Anxiety Attack and How Do You Stop One?

- Healthy Living
Sleepy Seniors Have Higher Health Risks

- Hospital Administration
DMC Names New CEO at Huron Valley-Sinai Hospital
- Neurology
Man paralyzed from neck down walks again, credits neurosurgeon from Michigan

- Cardiovascular
Detroit doctors help woman survive heart problem, continue teaching ballroom dancing
- Community
Wayne State running back hoping to impress at supplemental combine
- Community
Metro Detroit woman uses own health scare to inspire others for 'Go Red Day'

- Awards and Recognition
From Nurse to Detroit Medical Center CEO

- Awards and Recognition
Detroit Medical Center CEO Dr. Audrey Gregory on the perspective she brings to her role

- Community
New Chief Executive Officer at DMC - Paul W. Smith

- News and Articles
DETROIT MEDICAL CENTER TO BE OFFICIAL HEALTHCARE PROVIDER FOR DETROIT CITY FOOTBALL CLUB

- Community
Viral ‘skull breaker challenge’ circulating on social media puts children in danger

- Cardiovascular
Leading Heart Care Specialists Join DMC Heart & Vascular Team

- Hospital Administration
DMC CEO Dr. Anthony Tedeschi Announces Retirement

- Health and Wellness
DMC Puts Visitor Restrictions in Place to Fight the Flu

- Orthopedics
Outpatient Knee Replacement Surgery Now Offered at Huron Valley-Sinai Hospital

- Community
Sinai-Grace Guild Community Development Corporation Weatherizes Homes For Low-Income Seniors

- Awards
Huron Valley-Sinai Hospital Scores 'A' Rating From Leapfrog

- Orthopedics
Detroit Medical Center Teams With EXOS to Offer Sports Performance Services

- Awards
DMC Harper Hospital Ranked as One of Top in U.S.

- Community
DMC To Be Official Healthcare Provider For United Shore Professional Baseball League

- Awards
Dr. Anthony Tedeschi Recognized as One of the 50 Most Influential Clinical Executives by Modern Healthcare

- Maternity
Interactive Walking Path Created For Expectant Mothers At Huron Valley-Sinai Hospital

- Awards
Children’s Hospital of Michigan Ranks Among America’s Best in U.S. News & World Report 2019-20 Best Children’s Hospitals

- Health and Wellness
Flu Restrictions Lifted At Detroit Medical Center Hospitals

- Community
DMC Expands DLIVE Program To Serve More Trauma Patients

- Cardiac
Detroit Medical Center Welcomes New Leadership at Heart Hospital

- Community
Detroit Medical Center and Meharry Medical College Expand Medical Student Training Affiliation

New Non-Surgical Technique at Detroit Medical Center Benefits Dialysis Patients

Children’s Hospital of Michigan Launches First Neuro-Neonatal Intensive Care Unit in the State

- Community
Congresswoman Haley Stevens Visits Huron Valley-Sinai Hospital

- Community
Patty Jobbitt Named Chief Executive Officer of Rehabilitation Institute of Michigan

- Community
Detroit Medical Center to be Health Provider at U.S. Figure Skating Championships

- Community
Community Leader Appointed Executive Director at Sinai-Grace Guild Community Development Corporation

- Community
DMC Names New Leadership on Main Campus

- Community
DMC Strengthens its Commitment to Medical Education by Adding 79 New Residency Slots

- Community
Detroit Medical Center Announces Free Self-Parking at all of its Facilities
- Orthopedics
Patients come together for Mako Reunion at Huron Valley-Sinai Hospital Celebration of Mobility

- Oncology
Huron Valley-Sinai Hospital and Charach Family Honors Cancer Survivors at Annual “Just For You” Free Event

- Community
Detroit Medical Center Convenes Forum with Community Stakeholders to Assess Detroit’s Healthcare Needs

- Community
RUN with DMC smartphone application relaunches with new features

- Community
Detroit Medical Center and Wayne State University Physician Group Reach Agreement on Clinical Care

- Community
Rehabilitation Institute of Michigan Appoints Chief Nursing Officer

- Oncology
Sinai-Grace Hospital hosts Cancer Survivors' Day

- Community
Huron Valley-Sinai Hospital appoints Sheri Underwood as Chief Nursing Officer

- Oncology
Dr. Jason Wynberg of DMC Sinai-Grace Hospital helps local man suffering from Prostate Cancer on WDIV Channel 4

- Awards
DMC Hospitals Receive High Rankings in U.S. News & World Report

- Awards
Two Detroit Medical Center Hospitals Score Top Ratings from Leapfrog
- Neurology
DMC Sinai-Grace Ranks as One of the Best in the U.S. for Stroke Survival
- News and Articles
DMC Heart Hospital First in World to Treat Peripheral Artery Disease Through the Wrist

- Awards
U.S. News & World Report Names DMC Harper University Hospital Among Best in Michigan

- Community
Detroit Medical Center Announces Moves in C-Suite Positions

- Awards
The Children’s Hospital of Michigan Ranks Among America’s Best in U.S. News & World Report 2017-18

Eating Right for Your Kidneys

- Health and Wellness
Spice up Your Cooking

- Community
Monique Butler Appointed COO of the Children’s Hospital of Michigan
- Community
Because Hunger Doesn’t Take a Summer Vacation

- Neurology
If You’re Having a Stroke, What’s Your Action Plan?

- Neurology
The right ED Team and stroke diagnosis help a patient beat the odds

- Community
The Children’s Hospital of Michigan Appoints Chief of Physical Medicine and Rehabilitation

- Community
DMC RiverWalkers - A Fun and Healthy Walk Program for Seniors

- Health and Wellness
You’re not getting older. You’re getting better, with the DMC
- Awards
DMC Harper-Hutzel and Sinai-Grace Recognized by Leapfrog for Safety in High Risk Deliveries

- Awards
4 DMC Hospitals Receive Prestigious MPRO Governor’s Award of Excellence

- Maternity
DMC Hutzel Women's Hospital - Taking Care of Your Special Deliveries for over 150 years

- Health and Wellness
Here come those seasonal allergies
- Community
Partnership with AARP enhances DMC services to older adults
- Community
Pressurized Oxygen: The Miracle “Drug” for Wounds

- Orthopedics
Robotic Technology can Provide Knee Restoration

- Awards
Prestigious Award Recognizes Contributions to Improving Patient Safety

- Community
Detroit Medical Center Announces New CEO

- Neurology
DMC’s Children’s Hospital Names Award-Winning Clinician, Teacher, Researcher Chief of Pediatric Neurology

- Community
61 Day Challenge Kicks Off Fifth Year of Creating a Healthier Detroit

- News and Articles
DMC Harper-Hutzel Hospital Achieves Magnet Recognition

- Community
The Children’s Hospital of Michigan Earns EXCELLENCE THROUGH INSIGHT

- Surgery Center
DMC Heart Hospital Completes Highly Successful Complex High-Risk Indicated Interventional Procedure Course

DMC Heart Hospital Offers First-Of-Its-Kind Naturally Dissolving Heart Stent For Coronary Artery Disease

- News and Articles
DMC Heart Hospital VP Cindy Grines, MD, Selected for Ohio State University Alumni Achievement Award

- Community
Joel Keiper Returns to DMC as Regional Chief Business Development Officer

- Community
Veteran Detroit Medical Center CEO Dr. Reginald Eadie Named New Regional Chief Operating Officer

- Awards
Detroit Medical Center Achieves 10-Year Milestone on Nation’s "Most Wired" List

- Community
Cerebral Palsy Expert Appointed Chief of Neonatology at the Detroit Medical Center

Luanne Thomas Ewald Named Chief Executive Officer of DMC Children's Hospital of Michigan

- Awards
DMC's Hospitals Receive Get With The Guidelines-Stroke Silver Quality Achievement Award
Sign Up for Health Tips
Get our advice and upcoming events about weight, pain, heart and more.




